Solution Preparation System with Mixing Blending Dosing
Introduction
The integrated solution preparation system combines precision mixing, blending and dosing technologies for pharmaceutical liquid formulation. The system accurately incorporates Active Pharmaceutical Ingredients into solvents like WFI, achieving target concentrations through controlled stirring, heating, sterilization and filtration processes. Engineered for both sterile and non-sterile applications, this automated platform ensures consistent, high-quality pharmaceutical solutions.
System Classification
- General Formulation System: For small-molecule chemical prep (e.g., aqueous injections, lyophilized powder injections).
- Complex Formulation System: Handles advanced processes: suspensions, emulsions, liposomes, microspheres.
- Biological Formulation System: For biotech products (antibodies/vaccines/blood products/recombinant proteins) & auxiliary liquids (media/buffers).
Core Advantages
1) Modular Design - Cost-Saving & Efficient
Pre-tested modular equipment reduces installation costs, accelerates delivery, and ensures flexibility.

2) Fully Automated Operation - Smart & Compliant
1-click CIP/SIP with auto-detection for completion. Computerized recipe management, e-signature, e-records, and audit trail.
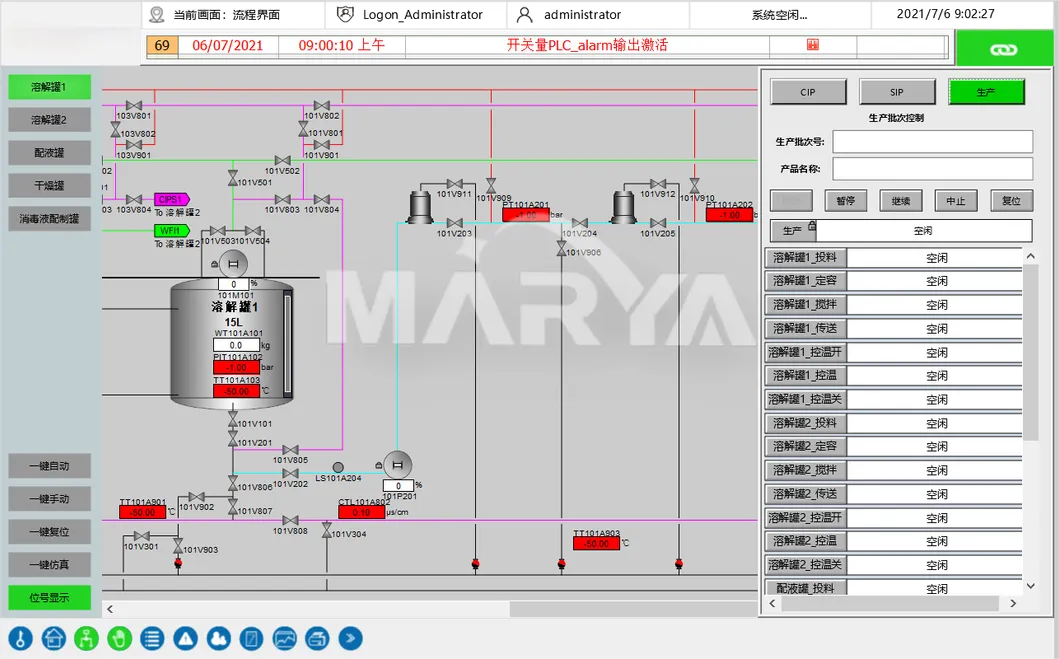
3) Process Control & Real-Time Monitoring
Critical parameter control (temp, DO, pH) with auto-alerts for deviations to ensure drug quality.

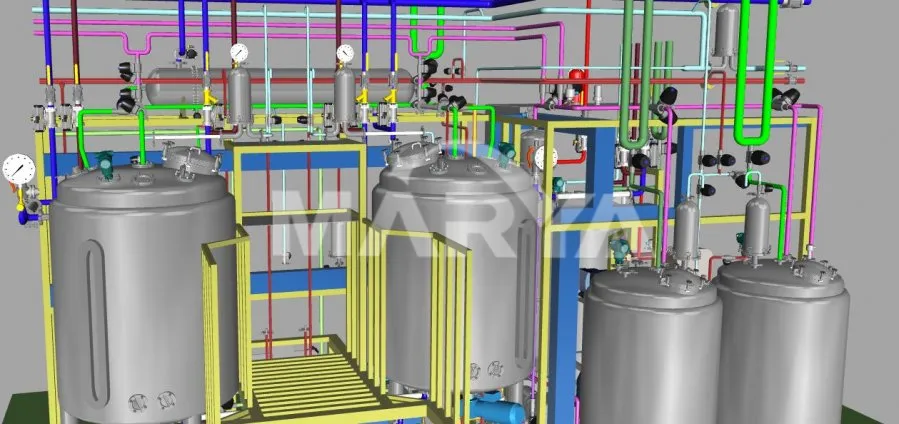
4) 3D Simulation & Ergonomic Layout
Pre-production 3D modeling optimizes piping, valve, and instrument placement for user-friendly operation.
Design Features
- Premium Material: 316L stainless steel tanks (2L-12,000L) with electrolytic polishing (Ra<0.4µm).
- Sterility Assurance: Sterile ventilation/exhaust filters and CIP spray balls for aseptic operations.
- Safety Systems: Integrated safety devices (burst disc/safety valve) and precision monitoring probes.
- Efficient Mixing: Magnetic stirrer (bottom) or mechanical stirrer (top) with frequency control.
- Filtration: Sterilization-grade PES filters resistant to 150°C and organic solvents.
Product Parameters
| Name |
Parameters |
| Working volume | 2~15000L |
| Material | 304, 316L, 904L, TA2 |
| Stirring type | Upper mechanical, lower magnetic stirring |
| Stirring speed accuracy | 0~450rpm ±3.0% |
| Temperature accuracy | -10~150ºC ±0.2ºC |
| Pressure accuracy | -0.01Mpa~0.06Mpa ±0.01MPa |
| Weighing accuracy | 3‰ |
| Power supply | 380V 50Hz |
| Protection levels | IP65 |
Frequently Asked Questions
Q1: What materials are used for the preparation tanks?
A: We primarily use high-grade 316L stainless steel for all contact parts, with options for 304, 904L, or TA2 based on specific requirements. All tanks feature electrolytic polishing (Ra<0.4µm).
Q2: Does the system support automated cleaning and sterilization?
A: Yes, the system features a "one-click" CIP (Clean-in-Place) and SIP (Sterilize-in-Place) operation with automatic detection to ensure the process is complete and compliant.
Q3: What range of working volumes can the system handle?
A: Our solution preparation systems are highly scalable, ranging from small 2L laboratory units up to large 15,000L industrial production modules.
Q4: How does the system ensure data compliance?
A: The control system includes computerized features such as recipe management, electronic signatures, electronic records, and a full audit trail to meet stringent pharmaceutical standards.
Q5: What types of stirring mechanisms are available?
A: We offer both upper mechanical stirring and lower magnetic stirring, both equipped with variable frequency speed regulation for precise control.
Q6: Are the filtration units resistant to high temperatures?
A: Yes, we utilize sterilization-grade PES filters that are resistant to temperatures up to 150°C and are compatible with various organic solvents.