














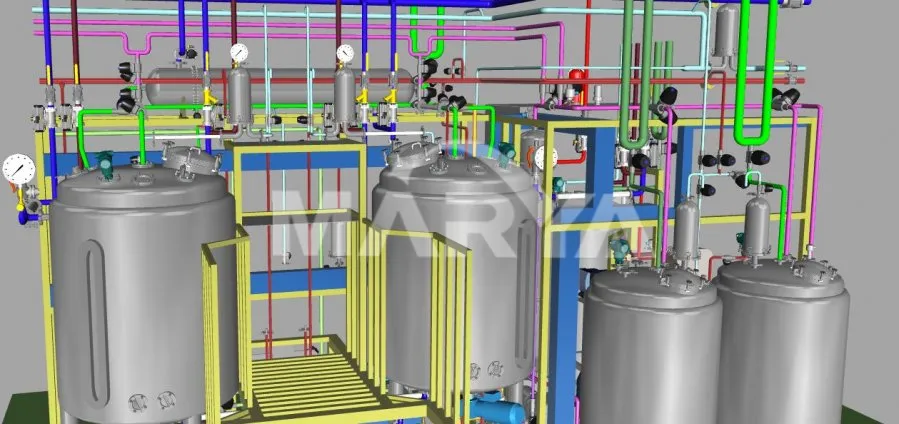
Vaccination Solution Mixer / Vaccine Blending Vessel / Inoculation Agitator / Formulation Mixing Tank with CIP/SIP System
Pharmaceutical dispensing systems are essential for preparing liquid formulations for various drugs. From raw materials to finished products—including antibiotics, vaccines, and biological preparations—the dispensing system ensures drug quality, efficacy, and safety.
1. General Formulation System: Used for small molecule chemical preparations such as aqueous injections and lyophilized powder injections.

2. Complex Formulation System: Designed for complex processes including suspensions, emulsions, liposomes, and microspheres.

3. Biological Formulation System: Applied in bio-engineering for antibodies, vaccines, blood products, and recombinant proteins, as well as culture media and purification buffers.


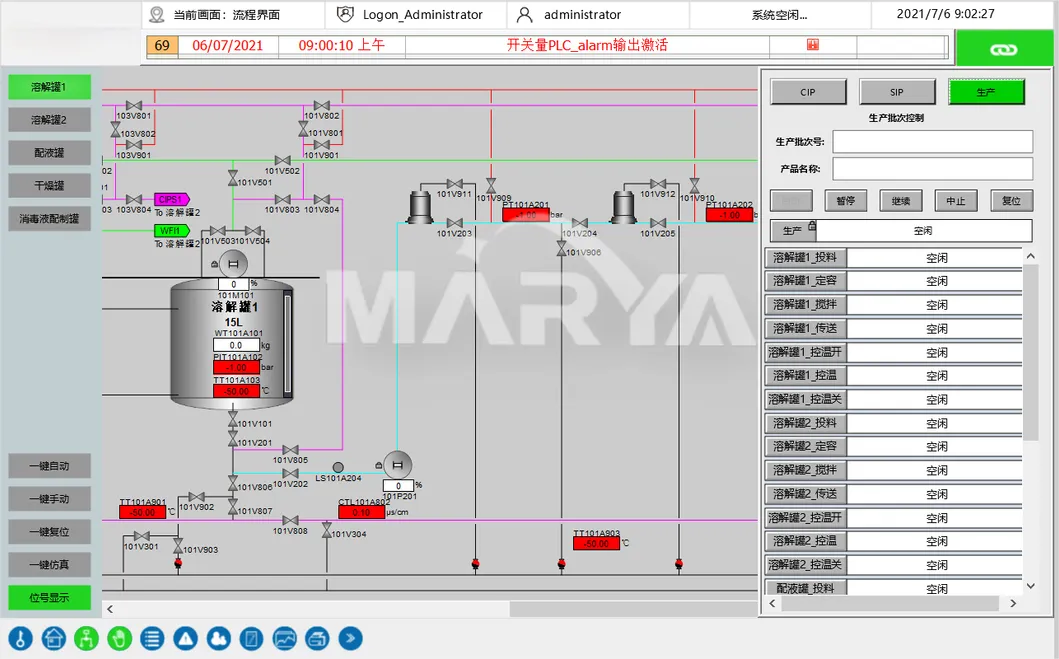

| Parameter Name | Specifications |
|---|---|
| Working Volume | 2 ~ 15000L |
| Stirring Speed Accuracy | 0 ~ 450rpm (±3.0%) |
| Temperature Accuracy | -10 ~ 150ºC (±0.2ºC) |
| Pressure Accuracy | -0.01Mpa ~ 0.06Mpa (±0.01MPa) |
| Weighing Accuracy | 3‰ |
| Power Supply | 380V 50Hz |
| Protection Levels | IP65 |












