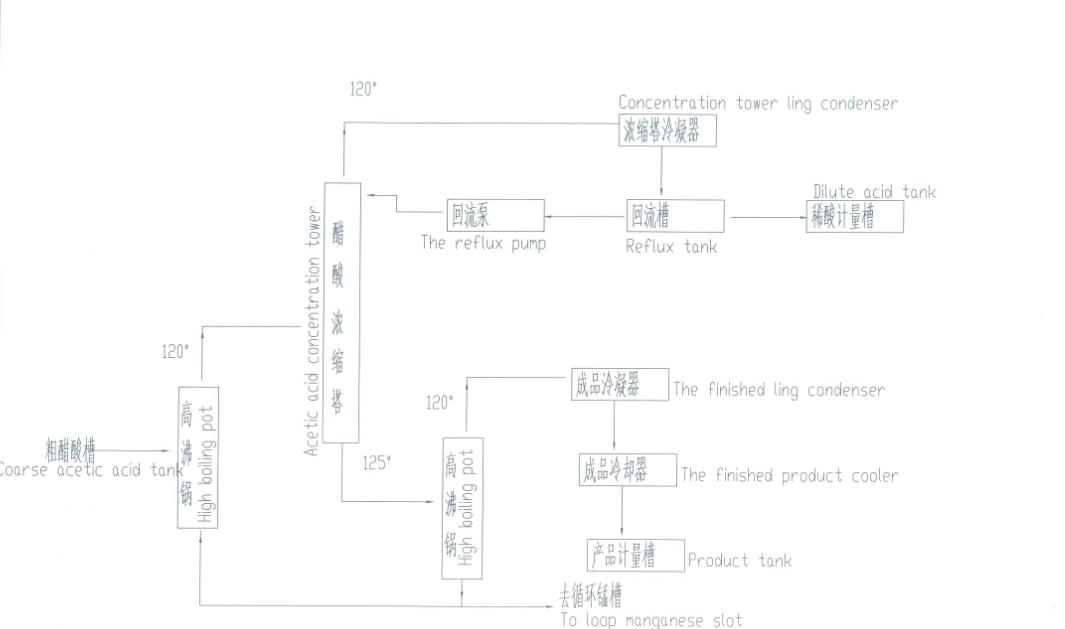
ℹ
Acetic acid (ethanoic acid) is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH. As the main component of vinegar (at least 4% by volume), it has been utilized since at least the third century BC and is also known as acetyl hydroxide (AcOH).
Primary Applications
- Chemical Production: Used for vinyl acetate monomer, acetic anhydride, and ester production.
- Medical Use: Applied in cervical cancer screening in developing regions.
- Food Industry: Used as a condiment and for pickling vegetables (4% to 8% concentration for table use).
- Industrial Solvent: Used as a polar protic solvent for recrystallization and TPA production (raw material for PET).
Acetic acid is produced through both synthetic methods and bacterial fermentation. Industrial production predominantly relies on the carbonylation of methanol (75%), while biological routes account for about 10%, primarily for food-grade vinegar.
1. Methanol Carbonylation
The most common industrial process involving the reaction of methanol and carbon monoxide:
CH3OH + HI → CH3I + H2O
CH3I + CO → CH3COI
CH3COI + H2O → CH3COOH + HI
Modern facilities utilize the iridium-catalyzed Cativa process, which is more efficient and environmentally friendly than older rhodium-catalyzed methods.
2. Acetaldehyde Oxidation
Previously a dominant technology, this method involves the oxidation of acetaldehyde derived from acetylene or light naphtha components.
2 C4H10 + 5 O2 → 4 CH3CO2H + 2 H2O
3. Ethylene Oxidation (Wacker Process)
Ethylene can be converted to acetic acid in a single-stage process using palladium catalysts. This method is competitive for smaller-scale production plants.
What is the common concentration of acetic acid in vinegar?
Table vinegar typically contains 4% to 8% acetic acid by volume, while commercial pickling solutions may use higher concentrations.
What are the main industrial uses of acetic acid?
The largest industrial use is in the production of vinyl acetate monomer (VAM), followed by acetic anhydride and ester production. It is also a critical solvent for TPA production.
How is industrial acetic acid primarily manufactured?
Approximately 75% of industrial acetic acid is produced via methanol carbonylation, specifically through the Monsanto or Cativa processes.
Can acetic acid be used in medical applications?
Yes, it is effectively used as a screening tool for cervical cancer in various developing regions worldwide.
Is biological fermentation still important for acetic acid production?
Yes, while it accounts for only 10% of global production, many food purity laws require vinegar used in food products to be of biological origin.
How can acetic acid be purified?
Acetic acid can be purified through fractional freezing. In an ice bath, pure acetic acid precipitates out while water and other impurities remain in liquid form.