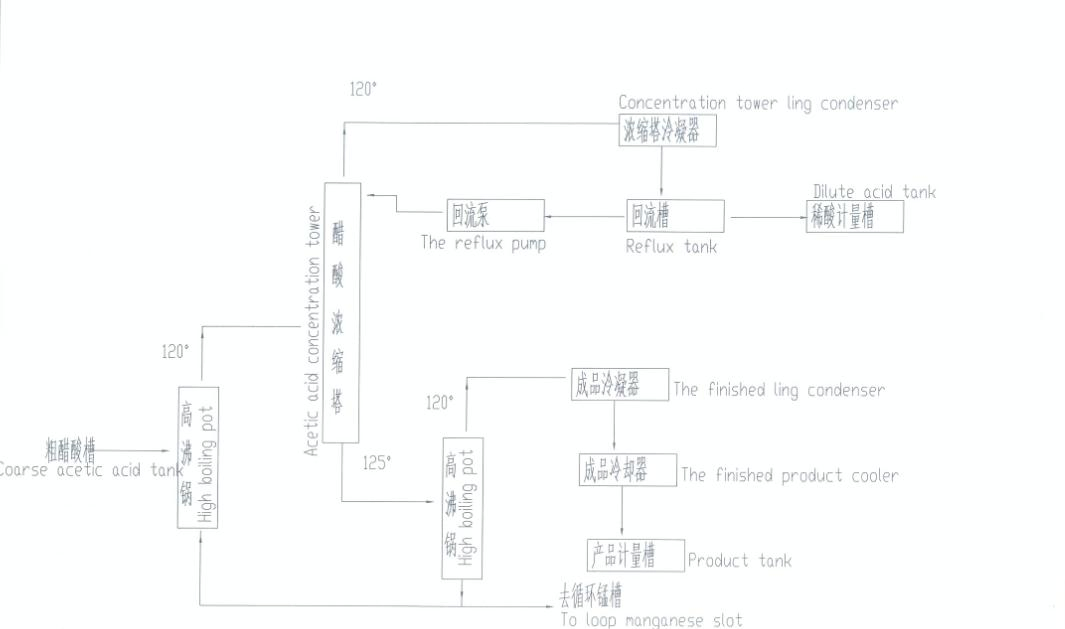
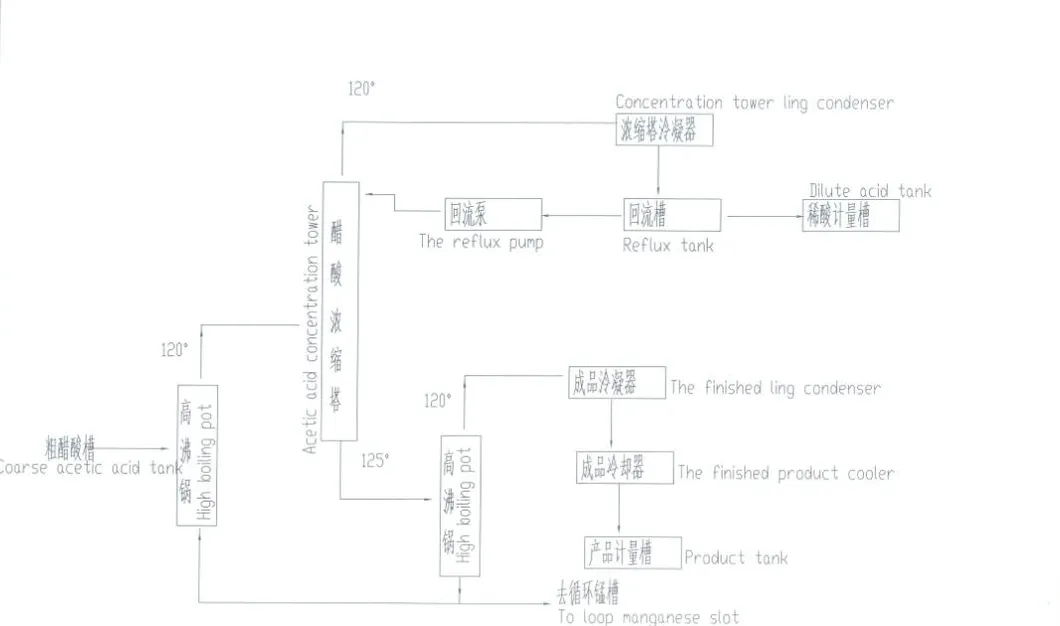
Product Description
Acetic acid, systematically named ethanoic acid, is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water. It has been used, as a component of vinegar, throughout history from at least the third century BC. Acetic acid is also known as acetyl hydroxide (AcOH).
professional automatic acetic acid making line venigar making machine
Acetic acid Use
Acetic acid is a chemical reagent for the production of chemical compounds. The largest single use of acetic acid is in the production of vinyl acetate monomer, closely followed by acetic anhydride and ester production. The volume of acetic acid used in vinegar is comparatively small.
- Medical: Acetic acid is used as part of cervical cancer screening in many areas in the developing world. The acid is applied to the cervix and if an area of white appears after about a minute the test is positive.
- Food: Vinegar is used directly as a condiment, and in the pickling of vegetables and other foods. Table vinegar tends to be more diluted (4% to 8% acetic acid), while commercial food pickling employs solutions that are more concentrated.
Use as solvent
As a polar protic solvent, acetic acid is frequently used for recrystallization to purify organic compounds. Acetic acid is used as a solvent in the production of terephthalic acid (TPA), the raw material for polyethylene terephthalate (PET). In 2006, about 20% of acetic acid was used for TPA production.
professional automatic acetic acid making line venigar making machine
Production
Acetic acid is produced industrially both synthetically and by bacterial fermentation. About 75% of acetic acid made for use in the chemical industry is made by the carbonylation of methanol. The biological route accounts for only about 10% of world production, but it remains important for the production of vinegar because many food purity laws require vinegar used in foods to be of biological origin.
Acetic acid can be purified via fractional freezing using an ice bath. The water and other impurities will remain liquid while the acetic acid will precipitate out. As of 2003-2005, total worldwide production of virgin acetic acid was estimated at 5 Mt/a. Global production has increased from 10.7 Mt/a in 2010 to 17.88 Mt/a in 2023.
Methanol carbonylation
Most acetic acid is produced by methanol carbonylation. In this process, methanol and carbon monoxide react to produce acetic acid. The process involves iodomethane as an intermediate and occurs in three steps:
- CH3OH + HI → CH3I + H2O
- CH3I + CO → CH3COI
- CH3COI + H2O → CH3COOH + HI
Two related processes exist: the rhodium-catalyzed Monsanto process, and the iridium-catalyzed Cativa process.
Acetaldehyde oxidation
Prior to the commercialization of the Monsanto process, most acetic acid was produced by oxidation of acetaldehyde. The acetaldehyde can be produced by hydration of acetylene. Light naphtha components are readily oxidized by oxygen or air to give peroxides, which decompose to produce acetic acid:
2 C4H10 + 5 O2 → 4 CH3CO2H + 2 H2O
Ethylene oxidation
Acetaldehyde may be prepared from ethylene via the Wacker process, and then oxidised. Showa Denko commercialised a cheaper single-stage conversion of ethylene to acetic acid using a palladium metal catalyst:
C2H4 + O2 → CH3CO2H
professional made automatic acetic acid production line venigar making machine line
Frequently Asked Questions
What is the chemical formula of Acetic Acid?
The chemical formula for acetic acid is CH3COOH, also commonly represented as C2H4O2.
What are the primary industrial uses of Acetic Acid?
The largest industrial use is for the production of vinyl acetate monomer, followed by acetic anhydride and ester production. It is also used as a solvent for TPA production.
How is Acetic Acid used in the food industry?
In the food industry, it is primarily used as vinegar (4% to 8% concentration) for condiments and pickling vegetables.
What is Methanol Carbonylation?
It is the most common industrial method for producing acetic acid, where methanol and carbon monoxide react using a catalyst like rhodium or iridium.
How is Acetic Acid purified?
It can be purified through fractional freezing using an ice bath, where the pure acetic acid precipitates out while impurities remain in liquid form.
What are the medical applications of Acetic Acid?
It is used in developing regions for cervical cancer screening; the acid is applied to the cervix, and a positive result is indicated by the appearance of a white area.